CFCs are a course of man-made compounds that were already broadly utilized in a wide extend of objects, including discuss conditioners, fridges, vaporized splashes, and fire quenchers. Carbon, chlorine, and fluorine combine to create CFCs. They are non-flammable, colorless, and odorless gasses.
Introduction
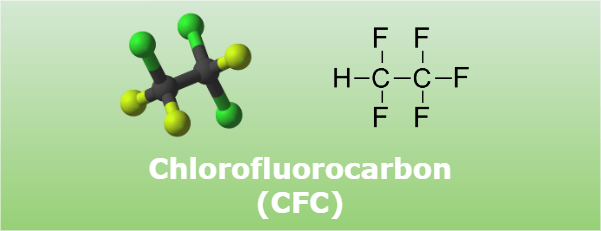
Chlorofluorocarbons (CFCs) are a group of synthetic organic compounds primarily made up of carbon, chlorine, and fluorine atoms. They were first introduced in the early 1930s and gained widespread use due to their remarkable stability, low toxicity, and non-flammability.
Initially celebrated for their safety and versatility, CFCs found applications in various industries, becoming integral to products such as refrigerants, propellants, foaming agents, and solvents. They played a crucial role in revolutionizing refrigeration, air conditioning, and aerosol technology, shaping modern living standards.
However, with time came a startling revelation: CFCs were severely damaging the Earth’s ozone layer. The ozone layer, situated in the stratosphere, acts as a shield, absorbing the majority of the sun’s harmful ultraviolet (UV) radiation. CFCs, once released into the atmosphere, would eventually rise to the stratosphere and release chlorine and bromine upon exposure to UV radiation. These elements would catalytically break down ozone molecules, causing significant ozone depletion.
The detrimental impact of CFCs on the ozone layer led to international concern, culminating in the Montreal Protocol on Substances that Deplete the Ozone Layer, an agreement signed in 1987. This treaty aimed to phase out the production and use of ozone-depleting substances, including CFCs, to protect the ozone layer and mitigate potential environmental and health hazards.
Chemical Structure and Properties
Chlorofluorocarbons (CFCs) are a family of man-made chemical compounds composed of carbon, chlorine, and fluorine atoms. Their structure resembles a carbon backbone surrounded by chlorine and fluorine atoms. Let’s dive into their chemical structure and understand their distinctive properties.
Chemical Structure:
CFCs have a general structure of CₓClᵧFᵨ, where x, y, and ρ denote the number of carbon, chlorine, and fluorine atoms, respectively. The carbon backbone is typically in the form of a linear or branched alkane (saturated hydrocarbon) structure.
Key Properties:
| Property | Description |
|---|---|
| Stability | Highly stable and non-reactive under normal environmental conditions, contributing to long atmospheric lifetime. |
| Boiling Points | Low boiling points, e.g., CFC-12 (dichlorodifluoromethane) boils at -29.8°C, ideal for refrigerants and propellants. |
| Toxicity | Generally considered to have low toxicity, enhancing safety for human handling and various applications. |
| Flammability | Non-flammable, ensuring safety in applications like refrigeration and air conditioning. |
| Ozone Depletion Potential (ODP) | High ODP values, contributing significantly to the depletion of the ozone layer. |
| Global Warming Potential (GWP) | High GWP, indicating their potency in trapping heat in the atmosphere and contributing to global warming. |
Uses and Applications
Chlorofluorocarbons (CFCs) found their way into numerous applications due to their exceptional properties. These compounds, once hailed for their versatility, were an integral part of modern living. Let’s uncover the diverse range of uses and applications that made CFCs ubiquitous.
- Refrigeration and Air Conditioning: CFCs revolutionized the refrigeration and air conditioning industry. Compounds like CFC-12 (dichlorodifluoromethane) were widely used as refrigerants, efficiently transferring heat and enabling temperature control in various cooling systems.
- Foam Blowing Agents: CFCs were crucial in creating foams for insulation in refrigerators, buildings, and vehicles. As blowing agents, they played a vital role in expanding and forming the foam structure, providing insulation properties.
- Aerosol Propellants: CFCs were extensively used as propellants in aerosol cans for products like deodorants, hair sprays, and insecticides. They facilitated the spray mechanism, ensuring the even and controlled release of the product.
- Solvents: CFCs served as effective solvents in various applications, including cleaning electronic components, precision cleaning in manufacturing, and as carrier fluids in various processes.
- Fire Extinguishers: Certain CFC compounds were used in fire extinguishers, contributing to fire safety measures by suppressing flames in case of fires.
- Medical Inhalers: CFCs were employed as propellants in inhalers used for delivering medications to individuals with respiratory conditions, such as asthma.
- Industrial and Commercial Applications: CFCs were used in a range of industrial applications, including as coolants in freezers, vending machines, and water coolers, as well as in manufacturing processes that required temperature regulation.
Environmental Impact
| Environmental Impact | Description |
|---|---|
| Ozone Layer Depletion | CFCs significantly contribute to ozone layer thinning. The release of chlorine and bromine atoms breaks down ozone molecules in the stratosphere. |
| Global Warming | CFCs possess a high Global Warming Potential (GWP), trapping heat in the atmosphere and contributing to global warming. |
| Amplified Climate Change Effects | The combination of ozone layer depletion and global warming creates a harmful synergy, intensifying the effects of climate change. |
| Health Implications | Ozone layer depletion due to CFCs can lead to increased UV radiation, causing health issues like skin cancers, cataracts, and immune system suppression. |
| Regulatory Responses | International agreements, notably the Montreal Protocol, aim to phase out CFCs, highlighting the proactive response to mitigate their environmental impact. |
Regulations and Phasing Out
In the face of the environmental havoc caused by Chlorofluorocarbons (CFCs), the international community united to address this critical issue. The main focus was on regulating the production, consumption, and eventual elimination of CFCs to restore the ailing ozone layer.
- Montreal Protocol: The Montreal Protocol on Substances that Deplete the Ozone Layer, adopted in 1987, stands as a monumental global treaty. Its primary objective is to phase out the production and use of ozone-depleting substances (ODS), including CFCs. The treaty gained widespread support and ratification, showcasing a unified front against the environmental threats posed by these compounds.
- Amendments and Adjustments: Over the years, the Montreal Protocol has been strengthened through several amendments and adjustments. These measures have accelerated the phase-out schedules for CFCs and related compounds, pushing for a faster transition to ozone-friendly alternatives. The amendments have demonstrated the global commitment to expedite the healing of the ozone layer.
- Successes and Challenges: The Montreal Protocol’s effectiveness is evident in the significant reduction of CFC production and consumption worldwide. The successful phase-out of CFCs in many countries has contributed to the recovery of the ozone layer. However, challenges persist, such as illegal trade, inadequate enforcement in some regions, and the need for ongoing education and advocacy to ensure complete compliance.
- Future Outlook: The proactive measures taken under the Montreal Protocol provide a blueprint for addressing global environmental concerns. It emphasizes international collaboration, scientific research, and a shared sense of responsibility. As we move forward, the lessons learned from regulating CFCs serve as a beacon of hope for effectively addressing emerging environmental challenges and nurturing a sustainable planet.
Alternatives to CFCs
| Refrigerant | Composition | Environmental Impact |
|---|---|---|
| Hydrochlorofluorocarbons (HCFCs) | Contain carbon, hydrogen, chlorine, and fluorine. | Lower ozone depletion potential than CFCs but being phased out due to global warming impact. |
| Hydrofluorocarbons (HFCs) | Contain carbon, hydrogen, and fluorine. | No ozone depletion potential but have a high Global Warming Potential (GWP). |
| Hydrocarbons (HCs) | Natural compounds like propane and isobutane. | Negligible impact on the ozone layer and low GWP. Widely used in refrigeration and air conditioning. |
| Ammonia (NH3) | Composed of nitrogen and hydrogen. | Zero ozone depletion potential and low GWP. Utilized in industrial and commercial refrigeration. |
| Carbon Dioxide (CO2 or R-744) | Pure carbon dioxide. | No ozone depletion potential, GWP of 1, making it extremely environmentally friendly. Used in various refrigeration and heat pump applications. |
| Air | Natural air composed of nitrogen, oxygen, and traces of other gases. | Most environmentally benign option with no ozone depletion or global warming concerns, although efficiency may not match other alternatives. |
Health and Safety Concerns
Health Concerns:
- Respiratory Irritation: Exposure to CFCs can cause irritation of the respiratory tract, leading to symptoms such as coughing, shortness of breath, and a sore throat.
- Skin and Eye Irritation: Contact with CFCs can irritate the skin and eyes, resulting in redness, itching, and discomfort.
- Neurological Effects: Prolonged or high-level exposure to CFCs may have adverse effects on the nervous system, potentially leading to headaches, dizziness, and in severe cases, unconsciousness.
- Cardiovascular Effects: Some studies suggest a possible link between exposure to CFCs and cardiovascular issues, including irregular heartbeat and changes in blood pressure.
- Reproductive and Developmental Effects: Research indicates that exposure to certain CFCs could have reproductive and developmental effects, although more research is needed to understand the extent of these effects.
Safety Concerns:
- Flammability Risk: Although CFCs themselves are non-flammable, they can decompose at high temperatures, releasing hazardous gases and potentially contributing to fire hazards.
- Chemical Reactivity: CFCs can react with certain chemicals, leading to the formation of toxic or harmful byproducts. This necessitates cautious handling and storage practices.
- Environmental Impact: CFCs are significant contributors to ozone layer depletion and global warming, making their safe disposal and usage critical for protecting the environment.
Conclusion
Chlorofluorocarbons (CFCs), once celebrated for their versatility and wide-ranging applications, turned out to be a cautionary tale in the history of environmental science and industrial practices. Their widespread use during the 20th century had severe and unintended consequences on the Earth’s protective ozone layer and climate.
In conclusion, the story of CFCs teaches us about the critical importance of thoroughly understanding the potential environmental impacts of new technologies before widespread adoption. It also demonstrates the effectiveness of global cooperation and proactive measures in mitigating environmental damage and transitioning to greener, more responsible practices.
FAQs
Chlorofluorocarbons (CFCs) are synthetic compounds made up of chlorine, fluorine, and carbon atoms. They were commonly used in the past in a variety of applications, including refrigeration, air conditioning, aerosol propellants, and foam-blowing agents.
CFCs were popular due to their stability, non-flammability, and excellent thermal properties, making them ideal for various applications. They were considered safe for a long time until their harmful effects on the ozone layer and the environment were discovered.
When released into the atmosphere, CFCs can break down due to solar radiation, releasing chlorine and bromine atoms. These atoms can catalytically destroy ozone molecules in the stratosphere, leading to the thinning of the ozone layer, also known as ozone depletion.
CFCs have a significant environmental impact, including ozone layer depletion and contribution to global warming. They have a high ozone depletion potential (ODP) and a high global warming potential (GWP), making them detrimental to the environment.
Read Also
Related posts:
- AMC Full Form: Benefits, Components, Needs, Advantage
- ORS Full Form: Dehydration, Myths, Flavors, Varieties & Facts
- PCC Full Form: Importance, Types, Application Process
- PAN Full Form: Legal Provisions, Regulations,
- BRB Full Form: Productive, Routine, Distractions
- MCD Full From: Introduction, Responsibility, Challenges
- CT Scan Full Form: Scans, price, Advantages
- USA Full Form: History, Economics,Technology, culture