- In order for plants to grow healthily, pH is crucial.
- The pepsin enzyme, which aids in the digestion of protein in diet, must be activated by pH.
- The human body is impacted by a change in the blood’s pH level.
Knowing the pH of various chemicals and other elements that we utilize on a regular basis is crucial. According to the pH scale, chemical components are typically classed as acidic, alkaline.
Introduction About pH
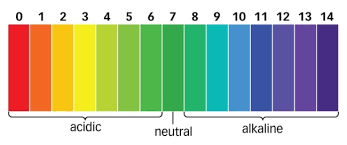
A fundamental idea that is essential to many aspects of our life is pH, or the value of hydrogen. It is a way to gauge a solution’s acidity or alkalinity and shows how many hydrogen ions (H+) are present. A pH scale of 0 to 14 is used, with 7 viewed as neutral. Acidic solutions have a pH under 7, and alkaline solutions have a pH besides 7.
Understanding pH is important because it affects many things, including industrial operations, human health, agriculture, and environmental sustainability. pH levels have a significant impact on everything in our environment, from the development of plants to the operation of our digestive systems.
What is pH Value?
The sharpness or alkalinity of a arrangement is decided by a solution’s pH esteem. It measures the sum of hydrogen particles (H+) display within the arrangement. A pH scale of to 14 is utilized, with 7 being respected as impartial. A pH esteem of 7 or higher recommends alkalinity, while one of 7 or less demonstrates causticity.
The pH scale is logarithmic, meaning that each entire pH unit speaks to a ten times distinction within the concentration of hydrogen particles. For case, a arrangement with a pH of 4 is ten times more acidic than a arrangement with a pH of 5, and a arrangement with a pH of 3 could be a hundred times more acidic than a arrangement with a pH of 5.
Substances can be categorized based on their pH values:
- pH 0-2: Strongly acidic
- pH 3-6: Weakly acidic
- pH 7: Neutral
- pH 8-11: Weakly alkaline (basic)
- pH 12-14: Strongly alkaline (basic)
pH is an vital parameter in different areas such as chemistry, science, natural science, and industry. It influences chemical responses, the behavior of substances, and the by and large wellbeing of natural frameworks. To degree pH, a pH meter or pH marker arrangements are commonly utilized.
Why pH Is Important for Daily Life
pH is important for daily life due to its significant impact on various aspects of our environment, health, and everyday activities. Here are some reasons why pH is important in our daily lives:
Wellbeing and Cleanliness: pH plays a pivotal part in keeping up the adjust of corrosive and antacid levels in our body. Diverse parts of the body have particular pH ranges that ought to be kept up for ideal working. For illustration, the pH of the skin, mouth, and stomach related tract influences our in general wellbeing and the development of advantageous microbes.
Drinking Water Quality: The pH of drinking water is imperative for its taste, security, and how successfully it can hydrate our bodies. Water that’s as well acidic or as well soluble can taste obnoxious and may moreover show the nearness of hurtful contaminants.
Farming: pH levels in soil impact the accessibility of fundamental supplements to plants. Diverse plants flourish completely different pH ranges. By understanding and altering soil pH, agriculturists and cultivators can optimize plant development and edit yields.
Cleaning and Family Items: Numerous family cleaning items, cleansers, and individual care things are defined to work inside particular pH ranges. The proper pH guarantees the adequacy of these items in evacuating soil, stains, and oils.
Nourishment and Cooking: pH may be a key figure in deciding the taste, surface, and security of nourishment. It influences the flavor and appearance of nourishments and can too affect the adequacy of cooking forms like heating and aging.
Swimming Pools and Aquariums: Keeping up the proper pH level in swimming pools and aquariums is basic for the wellbeing of both the water and the living beings living in it. Erroneous pH levels can lead to bacterial development, green growth blossoms, and stretch in oceanic life.
pH Value Influences Presence of Living Creatures
pH esteem undoubtedly features a noteworthy impact on the nearness and wellbeing of living animals, especially in sea-going situations and inside natural frameworks. Here’s how pH influences the nearness of living living beings:
Sea-going Life: pH levels in water bodies, such as lakes, streams, and seas, specifically affect oceanic life. Most sea-going living beings, counting angle, creepy crawlies, and microorganisms, have particular pH run inclinations. Sudden shifts in pH can stretch or indeed hurt these life forms. For case, on the off chance that the pH gets to be as well acidic (moo pH), it can lead to the discharge of poisonous metals from dregs, affecting oceanic life contrarily.
Coral Reefs: Coral reefs are exceedingly touchy biological systems that flourish in particular pH conditions. Sea fermentation, caused by expanded carbon dioxide levels within the environment, can lead to a diminish in sea pH. This drop in pH influences the ability of coral reefs to make and develop, because it disables the capacity of corals to construct their calcium carbonate skeletons.
Freshwater and Marine Life: Numerous sea-going species, from creatures of land and water to tiny fish, depend on particular pH ranges for survival. Changes in pH can disturb the accessibility of fundamental supplements and affect the behavior and regenerative designs of these life forms.
Creatures of land and water: Creatures of land and water are especially delicate to changes in pH due to their porous skin. Corrosive rain, which is rain with a moo pH due to contamination, can hurt creatures of land and water by influencing their skin, which they utilize for breath.
Microbial Life: Microorganisms, such as microbes and archaea, too have pH extend inclinations. Soil and water pH influences microbial communities and their capacity to break down natural matter, which has broader impacts on supplement cycling and environment wellbeing.
Plant Development: pH levels in soil impact supplement accessibility to plants. A few plants incline toward marginally acidic soil, whereas others flourish in soluble soil. The pH of the soil can influence the solvency of basic supplements like nitrogen, phosphorus, and potassium, which are pivotal for plant development.
Wetland Biological systems: Wetlands are basic environments that give homes for different species. pH impacts the accessibility of supplements and the chemical responses that happen in these situations. Changes in pH can affect the composition of plant and creature communities in wetlands.
Sea Biological systems: The pH of the seas has been diminishing due to expanded carbon dioxide levels, a marvel known as sea fermentation. This has suggestions for marine life, particularly living beings with calcium carbonate shells or skeletons, like mollusks and a few sorts of tiny fish.
pH Value in the Digestive System
pH plays a pivotal part within the stomach related framework, where it makes a difference encourage the breakdown of nourishment, absorption, and retention of supplements. Diverse parts of the stomach related tract have shifting pH levels that are optimized for particular capacities. Here’s how pH works totally different parts of the stomach related framework:
Mouth: The beginning step of absorption takes put within the mouth, where nourishment is broken down mechanically by chewing and blended with spit. Spit contains chemicals like amylase, which starts the assimilation of carbohydrates. The pH of spit is regularly around 6.5 to 7.5, which is marginally acidic to unbiased. This pH run makes a difference keep up chemical movement and start the breakdown of starches.
Stomach: The stomach could be a profoundly acidic environment with a pH extending from around 1.5 to 3.5. This moo pH is essentially due to the discharge of hydrochloric corrosive (HCl) by gastric organs.
Small Intestine: After taking off the stomach, the somewhat processed nourishment, known as chyme, enters the little digestive system. The pH within the little digestive system is somewhat soluble, extending from almost 7 to 8.5. This soluble environment is kept up by the emission of bicarbonate particles from the pancreas and bile from the liver.
Large Intestine: As nourishment moves through the expansive digestive tract, the pH remains moderately impartial, around 6.5 to 7.5. Whereas most absorption is total by the time nourishment comes to the huge digestive tract, a few remaining supplement assimilation happens here, in conjunction with water assimilation and the arrangement of feces.
Changes in pH Value Cause Tooth Decay
Vacillations in pH Levels Contribute to Dental Caries: This emphasizes that shifts in pH play a part in causing tooth rot.
Varieties in pH Values Lead to Cavities Arrangement: This highlights how changes in pH can result within the improvement of cavities.
pH Level Modifications Result in Dental Disintegration: This underscores that alterations in pH can lead to the disintegration of tooth finish.
Shifts in Sharpness Levels Can Trigger Tooth Disintegration: This indicates that changes within the level of sharpness can start the disintegration of teeth.
pH Changes Cause Tooth Debasement: This focuses out that modifications in pH can bring around the breakdown of tooth structure.
pH Changes Contribute to Dental Disintegration: This emphasizes that variations in pH can play a portion within the weakening of dental wellbeing.
Fluctuating pH Levels Lead to Tooth Breakdown: This proposes that changes in pH can result within the breakdown of teeth.
Modifications in pH Values Result in Tooth Harm: This underscores that changes in pH can cause hurt to teeth.
Frequently Asked Questions
pH is important in everyday life because it influences various aspects of our health, environment, and daily activities. It affects everything from the taste of our food and the effectiveness of cleaning products to the health of aquatic ecosystems and the growth of plants.
pH influences our bodies by keeping up the adjust of acids and bases in different frameworks. For case, the pH of our blood and stomach related tract influences supplement assimilation, protein movement, and the development of advantageous microbes. that’s why its important to keep in mind the pH importance in everyday life .
pH influences the effectiveness of skincare products and their compatibility with our skin. Skin has a natural pH that needs to be maintained for healthy functioning and to prevent issues like dryness and irritation.
pH affects the taste, texture, and safety of food. It can impact the way ingredients interact during cooking and influence the outcome of chemical reactions like fermentation and browning.
Soil pH affects nutrient availability to plants. Different plants thrive in different pH ranges, so adjusting soil pH helps optimize plant growth and crop yields.
Related posts:
- 7 Science Fiction movies on Prime for science students
- TOP 10 SOFTWARE DEVELOPMENT COMPANIES IN USA WHO EMPLOY GRADUATES
- Top 6 Institutes For Mathematics
- Career Choices In Health Care Sector
- Top 5 Delhi University Colleges For Science
- BSc Forensic Science
- Bachelors in Pharmacy as a career option
- Paramedical Courses 2023: Admission, Colleges, Scope